TEACHER BACKGROUND-DISSOLVED OXYGEN
Background:
Animals in a water environment require oxygen in order to breathe. Some of the free oxygen (oxygen gas, O2) in the water comes from photosynthesis as plants take up carbon dioxide dissolved in the water and release oxygen gas as a byproduct. Most of the free oxygen found the water comes from the atmosphere where water interfaces with air. Fish and invertebrates that live in the water get free oxygen by means of their gills. Gills are composed of abundant surface area where gases dissolved in water can diffuse in and out of the blood in the gills. Free oxygen diffuses into blood in the gills and carbon dioxide and other metabolic gases are expelled.
Water contains significantly less free oxygen compared to the atmosphere. Lake or river water usually contains less than 12 parts of free oxygen for every million parts of water molecules (12/1,000,000) or .000012 %. Air has 21 parts of free oxygen per hundred parts of atmosphere (21/100) or 21%. If water has less than 4-5 parts of oxygen per million parts of water, the animals have difficulty and can suffocate.
The amount of dissolved oxygen in water is affected by temperature. Cold water, with its slow moving molecules, holds more dissolved oxygen than warm water, whose faster moving molecules push some of the free oxygen atoms into the atmosphere. (This might seem at odds with how solids dissolve in water. Warm water can dissolve more sugar than cold water can. To help make the point with gases, however, think about soda. The bubbles in soda are dissolved gases. Cold soda is very bubbly. Warm soda cannot hold as much gas, so it goes flat.) Dissolved oxygen is also affected by wind-pushed waves and fast-flowing waters that increase opportunities for oxygen from the atmosphere to dissolve into the water. After the students complete their experiment, be sure to discuss how water temperature and the amount of water movement affect dissolved oxygen.
Preparation:
The test kits all require a water sample of a specified amount and the adding of several chemicals to the sample to allow the free oxygen molecules to be calculated. The simplest test kits are the CHEMette kits. You can purchase a CHEMette kit with 100 tests for $40 from CHEMetrics, Inc. The La Motte Company has a simple test that requires placing a tablet containing the necessary chemicals in a sample of water, watching the water change color, and comparing the color to a color chart. The kit costs about $20. Both tests are easy to use. See “Resources” below for addresses.
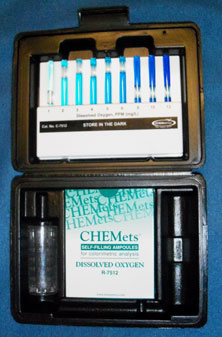
Resources:
Link to Chemetrics, Inc where you can order a catalogue http://www.chemetrics.com.au/content.php?ID=2
This link shows a picture of the oxygen test kit that is appropriate for students. The kits have 30 ampules and an ampule chart that helps students determine oxygen levels. http://www.chemetrics.com/Oxygen+%28dissolved%29
Link to LaMotte Company’s web site with information about ordering their catalogue and links to other resources. http://www.lamotte.com/pages/edu/index.htm
Procedure:
1. Explain Data Sheet #8 to the students and distribute them with clipboards to each group.
2. Have water samples from different sources in labeled containers available for the students to test. Each team can test a different water sample so students do not use too may ampules.
Discussion:
1. How does free oxygen get into the water? (Plants photosynthesize and give off oxygen as a byproduct. This oxygen gas dissolves in the water. Some oxygen gas mixes in from the air where the water and air touch. On windy days, or if the river is moving swiftly over rocks and water falls, even more oxygen mixes in from the air. It would be interesting to do the test in quiet waters at the headwaters of the river and again in swifter flowing water at another location. You could also test the oxygen level at the same site at different times of the year.)
2. Why is dissolved oxygen important to fish and other animals that live in the water? (They must have oxygen gas, just like other animals. Fish must have at least 4-5 parts of oxygen per million parts of water or they suffocate.)
3. Does the temperature of water affect the amount of oxygen gas in the water? (Yes, the colder the water, the slower the water molecules move, so the more free oxygen is able to stay in the water. As the temperature rises, water molecules move faster and bump some free oxygen atoms into the atmosphere.)
CHAPERONE CHEAT SHEET: DISSOLVED OXYGEN
Collect a water sample gently using a small pail. Do not agitate the water.
Open the dissolved oxygen kit. Remove the plastic measuring cup and fill it to 25 ml (the top mark).
Remove a vial from the white box in the kit. Place the pointed end in a bottom "corner" of the measuring cup, and then apply enough pressure to break the tip off. When the tip is broken, water from the cup will go into the vial.
Remove the vial from the cup. Flip the vial top over bottom twice to mix the contents. Wait two minutes.
Place the vial, pointed end up, next to the indicator vials and compare colors to see which vial is the closest match to the sample.
Record the reading in ppm (parts per million) on the data sheet under "oxygen" and the correct town or site number.
Water usually has less than 12 ppm dissolved oxygen. Most aquatic creatures need at least 5ppm dissolved oxygen or they have difficulty breathing.
STUDENT EXPERIMENTS----DISSOLVED OXYGEN TEAM
Overview:
In this experiment, your team will measure the amount of dissolved oxygen at your selected river location and learn what the measurement indicates about your site. Animals living in the water depend upon the dissolved oxygen to breathe. For example, fish must have 4 to 5 parts of oxygen per million parts of water or they suffocate. You will be measuring free oxygen that is dissolved in the water and available for animals to breathe. A measurement of eight to ten parts of oxygen gas per million parts of water means that the river is a healthy place for fish, mussels, and other water animals to live.
Focus:
How much oxygen is dissolved in the water at your river site? Is this site a good place for fish to live?
Materials:
___dissolved oxygen test kit
___several clean containers
___water from several different sources
___pencil
___data sheet and clipboard
Procedure:
Pre-field experiment:
1. Your teacher has set up labeled water samples from different sources in small buckets about 1/2 full.
2. The temperature group will take the temperature of each sample, record it, and tell you what it is. Write these readings on the chart that the teacher will provide for you on your data sheet.
3. Next, follow the directions for using the dissolved oxygen kit to determine how much oxygen is in the water sample you selected.
4. Record the amount of dissolved oxygen on your data sheet. Could a fish live in these waters? Why or why not?
Field experiment:
1. Collect a sample of water from your river location in the bucket. Fill the bucket about 2/3 full.
2. Submerge the small bottle from the CHEMette kit and fill it to the top line.
3. Put a vial containing the indicator chemical into the bottle, press down hard and break the tip of the vial.
4. Watch the water come into the vial and mix with the indicator chemical.
5. Leave the vial in for about 2 minutes and watch the color change from clear to blue.
6. When the color stops changing, place the vial in the “comparator” and compare colors to find the amount of oxygen in the water. Look at the number next to the color of the vial containing your sample. This is the number of oxygen molecules that are dissolved in each million molecules of water.
7. Write down the amount of dissolved oxygen on your data sheet. Dissolved oxygen is measured in parts per million (ppm).
Discussion:
1. We have much more oxygen in the atmosphere to breathe than fish have in water. We have about 21 parts per hundred and they have only a few parts per million. How can they live with so little oxygen?
2. Was the river the same temperature at each site that you measured? Were the dissolved oxygen levels the same at each site that you measured?
3. Does cold water hold more oxygen gas than warmer water? Why?
4. Most trout prefer to live in colder waters. What might be one reason for this? (Clue: Does your experiment help you answer this question?)

